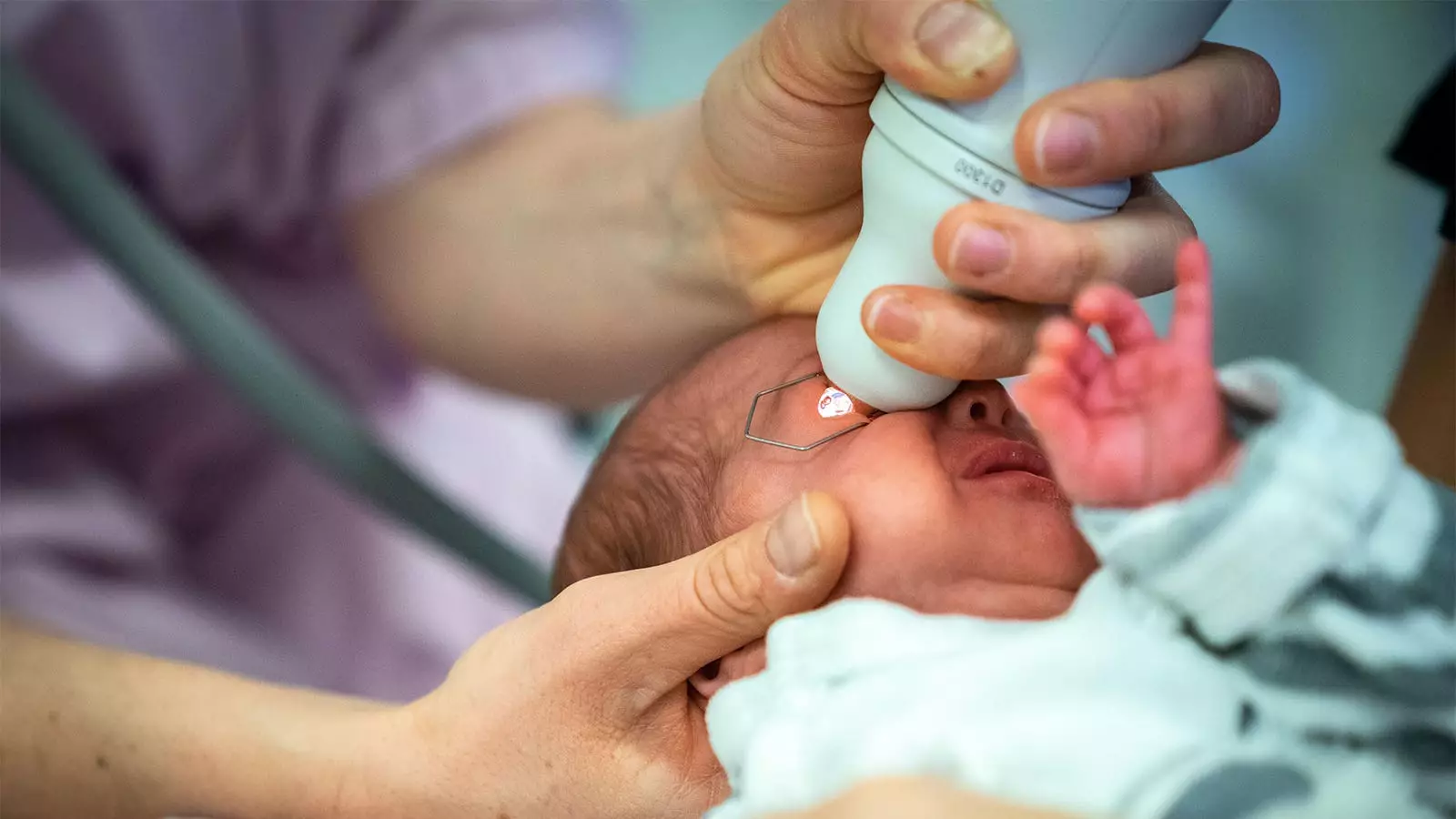
Retinopathy of prematurity (ROP) is a potentially serious eye condition that primarily affects preterm infants, particularly those with very low birth weights. Early detection through routine eye examinations is critical to preventing severe visual impairment or blindness in these vulnerable patients. Standard practice for facilitating these examinations involves applying mydriatic eye drops, which help dilate the pupils. However, the use of these conventional drops has raised concerns regarding systemic adverse effects, particularly in fragile preterm infants, whose immature organ systems may struggle to cope with the pharmacological impact of standard doses.
A recent randomized trial highlighted by Asimina Mataftsi, MD, PhD, from Aristotle University of Thessaloniki, has opened the door for an alternative approach using mydriatic microdrops. In this study, the effectiveness of these microdrops was compared directly against standard mydriatic drops. The findings were significant, revealing that not only were microdrops noninferior to standard drops, but they also exhibited superior mydriatic efficacy at the critical 45-minute mark. The mean difference measured was 0.12 mm, with a robust statistical significance (P=0.008).
Moreover, at 90 and 120 minutes post-instillation, the microdrops maintained their efficacy, with the study establishing that while their performance was on par with conventional methods, the adverse effects associated with standard drops were notably reduced. This outcome serves to underscore the potential benefits of microdrops as both an effective and safer alternative for this at-risk population.
One of the pivotal strands of the research involved monitoring adverse health outcomes among infants following the use of standard dilating drops. The study revealed concerning trends—lower oxygen saturation levels at the 45 and 90-minute marks after administering standard drops, with significant correspondences to adverse cardiorespiratory implications (P=0.03, P=0.04 respectively). Furthermore, the incidence of hypertensive episodes within 24 hours was markedly higher among those treated with standard drops, hinting at potentially severe systemic responses to these traditional mydriatics.
Such findings echo sentiments expressed in previous literature that indicates the heightened risks that mydriatics may pose to preterm infants. As discussed in a 2019 review, the association with cardiovascular, gastrointestinal, and respiratory complications necessitates a reevaluation of how mydriatics are utilized in this patient demographic. The compounded findings of adverse events following routine eye examinations are substantiated by anecdotal reports from NICU nurses, further emphasizing the clinical challenges associated with current practices.
The MyMiROPS (Mydriatics in ROP Screening) study conducted across a tertiary care center encompassed 83 preterm infants who met strict eligibility criteria, including gestational age and birth weight thresholds. The infants were randomly assigned to receive either microdrops or traditional drops, allowing for a controlled comparison of outcomes.
The trial’s methodology was comprehensive, with precise measurements taken of pupil diameter at three critical intervals post-administration using a customized measuring tool. Although the study had a rigorous framework, limitations were acknowledged, including a notable gap in safety outcome data for a portion of the patient sample. This gap will need to be filled by future investigations that could illuminate further the systemic exposure variations associated with both mydriatic methods.
While the findings of the MyMiROPS study present compelling evidence favoring the implementation of mydriatic microdrops for ROP screenings, caution and further inquiry are warranted. The implications of these results extend beyond immediate clinical practices; they highlight a pressing need for broader studies that can validate these findings in varied populations and clinical settings. Learning more about the systemic effects linked to both administration methods promises not only to improve the safety profile of ROP screenings but also to enhance overall neonatal care practices.
This novel study paves the way for innovative approaches to an age-old practice, challenging established protocols and advocating for a shift towards safer and equally effective mydriatic alternatives. The vulnerability of preterm infants must remain a central consideration in treatment approaches, ensuring that their care prioritizes not only effectiveness in screening but also their delicate physiological integrity.

Leave a Reply